By Brett Dworski
For as long as I can remember, I’ve been a short guy.
You could say that shortness—being vertically challenged—runs in my family, being that the tallest person in it, my Uncle Mike, is 5’10”.
Standing 5’6” is not the worst thing in the world. There are certainly more dire problems to have. Plus, what I lack in height, I make up for in bulk, weighing nearly 170 pounds, with an athletic build. What most people don’t know is that if it weren’t for a special treatment I had when I was younger, I could have been an entire foot shorter today.
I stopped growing at 3 years old. My parents, confused as to why their otherwise perfectly healthy toddler would just stop growing, sought answers. This led them to Stephen Duck, MD, an endocrinologist at NorthShore University HealthSystem. Duck ran some tests and concluded that I had growth hormone deficiency (GHD), which affects 1 out of 3,400 kids in the United States.
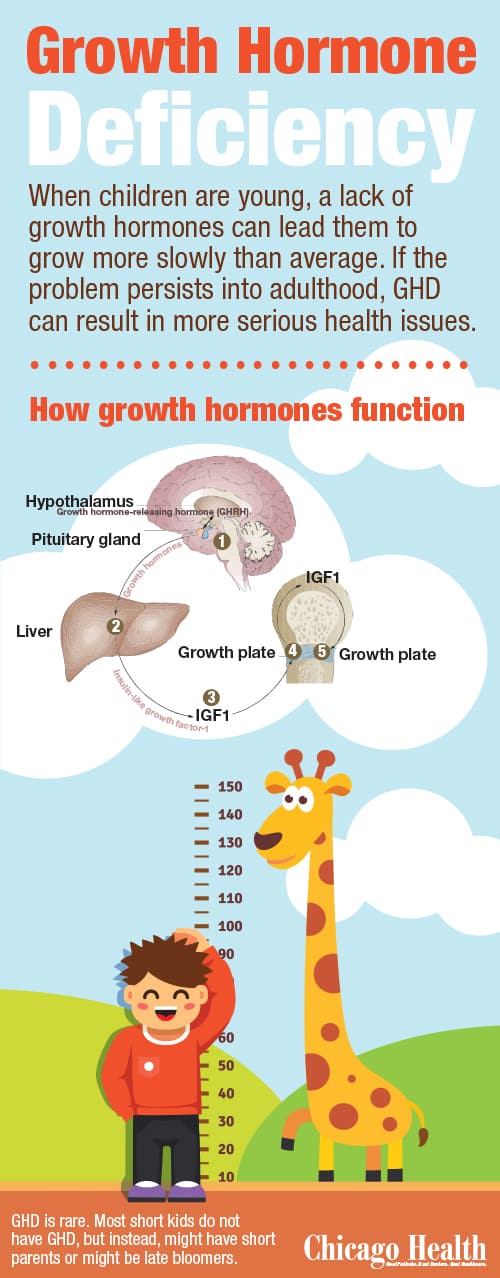
GHD is exactly what it sounds like: a deficiency in growth hormones. And growth hormones are needed to, well, grow. When children are young, a lack of these hormones leads them to grow more slowly than average. If the problem persists into adulthood, GHD can result in more serious health issues.
Growth hormones originate in the pituitary gland and travel through the bloodstream to the liver to create a protein called insulin-like growth factor-1, or IGF1. This protein provides necessary anabolic benefits. “It maintains cholesterol in a positive way—good cholesterol stays up while bad cholesterol stays down. It helps maintain muscle mass and keeps fat mass down. And lastly, it’s important in maintaining bone strength, integrity and mineralization,” Duck says.
As rare as GHD is in children, it’s even rarer in adults. Many kids grow out of it. Which is to say, testing for GHD is not perfect. Some kids who are treated for GHD during growth will eventually stop taking growth hormones without any problems. If the test were to be repeated, many of these kids would have normal tests, and it may be unclear whether they ever had a deficiency, says Siri Atma W. Greeley, MD, PhD, a pediatric endocrinologist at University of Chicago Medicine.
Permanent forms of GHD are often a result of anatomic or genetic problems or from tumors, Greeley says. Most short kids do not have GHD, but instead, might have short parents or might be late bloomers, he adds. But if a child isn’t growing, it’s important for him or her to be checked for medical problems that could affect growth, possibly including testing for GHD.
Growth Tests Coming Up Short
The problem is that testing is not definitive enough.
Diagnostic testing uses blood tests as well as descriptors, like skeletal maturation relative to birth, or how fast the child grows in comparison to age. It’s a descriptive model as opposed to a genetic one like tests used to define Down and Turner syndromes.
“The biggest problem facing pediatric endocrinology is the diagnosis of GHD. It’s not efficient,” Duck says.
GHD treatment can cost up to $40,000 a year, with treatments generally lasting through puberty. My treatment began at age 3 and lasted for 12 years. That’s a rough cost of $480,000. While some amount may be covered by insurance, Duck says, with increasing co-pays and deductibles, the burden can be onerous.
My parents don’t regret spending the money, as I’m sure many other parents wouldn’t. But that’s a lot of dough to fork over for treatment of a deficiency that isn’t life threatening. That’s why a better diagnosis is necessary to avoid unnecessary and costly treatment for those who are simply late bloomers.
The tests Duck gave me revealed that I was not lacking in growth hormones, but instead, incapable of sending out the hormone that releases the proper hormones. For growth hormones to head to the liver and make IGF1, the brain’s hypothalamus needs to send a growth hormone-releasing hormone (GHRH) to the pituitary gland.
I was lucky because back in 1998, when my treatment began, the FDA allowed a drug called GHRH to be administered. I was given the drug for two years, and during that time, I grew as expected.
“The amount of first-year growth depends on factors such as age when the treatment begins, the amount of milligrams given per dose and how often each dose is given,” Duck says.
I grew an average of 3.5 inches a year, but two years after my GHRH treatment began, the FDA took it off the market. Duck then gave me the standard GHD injections, and my growth remained steady. By the time I was 15, I was 5’5”. Every kid reaches a growth plateau, and that was mine. I was taken off the hormones and grew another inch.
Reaching to the Revolution
We don’t know whether I reached 5’6” instead of 4’6” because of the treatments. Maybe I was just a late bloomer. No one else in my family was treated for GHD. But because there are not yet the genetic testing capabilities to provide certain results, many parents are spending $40,000 a year to get their kids growing.
Duck says that growth hormone treatment nowadays is no different than it was when I was young. “The only difference,” he says, “is that they’ve placed more emphasis not on the treatment itself, but in making the diagnosis.”
A better genetic test is needed. “Right now, it’s very expensive, and we don’t yet know all the genes we want to test for,” he says.
By analyzing someone’s genetic composition, pediatric endocrinologists would be able to more accurately determine whether a kid has GHD or is just a late bloomer, thus saving families hundreds of thousands of dollars in unnecessary medical expenses. Duck says that genetic testing will lead to kids getting treated sooner and better, and that it will make the entire process a better experience for patients and their families.
“Genetic analysis,” Duck says, “is going to revolutionize every aspect of medicine.”