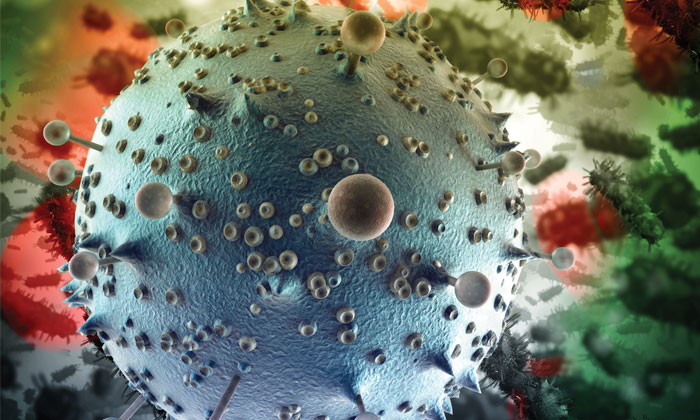
Science indicates that cancer stem cells play a role in the recurrence of tumors; the challenge now is to find them
In science, controversy arises from uncertainty. The role of cancer stem cells being used to treat and prevent tumors is still a far cry from certain. But new studies published early last month in the journals Nature and Science add to the mounting evidence supporting the theory.
Stem cells are those that divide to become either new stem cells, which will continue to divide, or become a completely new type of cell with a more specialized function such as a muscle cell, brain cell or red blood cell, according to the National Institutes of Health. Two types exist; embryonic stem cells and adult stem cells. In adults, these cells act as a repair system for the body by replenishing tissues. This can include the tissues of cancerous tumors.
“The concept of a cancer stem cell is a very useful one because it would explain some of the really confounding things that happen in cancer patients,” says Dr. Donald Braun, vice president of clinical research at Cancer Treatment Centers of America (CTCA) in Zion.
For scientists and doctors, the return of a tumor 10 or 15 years after it has seemingly been completely eradicated is no less than confounding. For a woman facing another battle with breast cancer, it’s much more than that.
To the thinking of Braun and others in the field, the best way for them to help patients is to find the root of the tumor’s recurrence. “When a breast cancer recurs after 20 years, it’s probably not due to a second, identical genetic mutation. It’s more probable that there was a precursor cell that’s been sitting around dormant that became reactivated for some reason.
“We know that tissues that are not malignant, such as the white and red blood cells in the circulation, are renewed by stem cells,” he says. Embryonic stem cells are the ultimate mothership from which all the tissues of the body descend. But there are stem cells present in the adult body that are specifically geared toward reproducing a certain set of tissues.
“Contained within [a stem cell] is the potential for all of the downstream forms of a tissue,” says Braun. “It’s just a question of turning on genes in that stem cell to get [it] to look like [a] specialized [cell].”
Braun explains that stem cells can also be transplanted from one environment to another, as is seen in certain cardiac or orthopedic patients, where fat cells are strategically relocated and are able to learn new instructions and begin to produce a new set of specialized cells. “Cancer replicates that behavior, but does it in a way that is not particularly civilized,” he says.
Unlike the helpful fat cell chipping in to aid the heart or bone, the downstream lineages of a cancer cell include the capacity to invade tissues, resist signals that tell the cell to die (which normal cells will respond to), metastasize and promote revascularization (abnormal growth of blood vessels).
The current approach to treatment focuses on these downstream effects and how best to fight them. But understanding where the process begins is key to eradicating cancer for good.
And that’s where the cancer stem cell theory becomes forward-looking. Based on what we know, the theory seems sound, and the observable evidence continues to build. But as to where these insidious cancer stem cells come from, how we come to carry them, at what point in our lives and why? “Those are the kinds of questions [that] lead to Nobel prizes,” says Braun. If, of course, we can answer them. [email_link]