An adventure through the evolving science of organ transplantation
By David Himmel
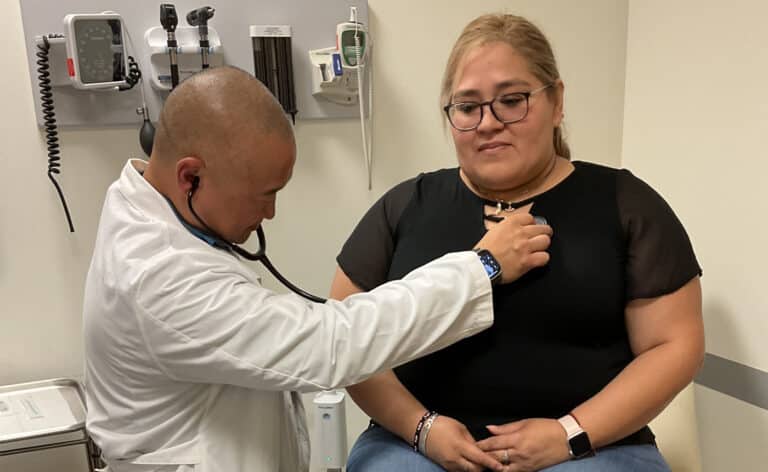
It is late morning in early May 2014. Ozzie Rivero’s pager buzzes. The message reads: “heart offer.” Rivero takes action. Within the next 24 hours, he will be part of a harried effort to retrieve a still-beating heart from a deceased donor’s body and get it into the recipient’s waiting body before time runs out.
For Rivero, the organ procurement coordinator at the University of Chicago Medical Center (UCMC), days like these are commonplace. If it isn’t a heart, it’s a kidney, a liver, a pancreas or lungs.
The Gift of Hope Organ and Tissue Donor Network, the not-for-profit organ procurement organization that coordinates transplants in Illinois and northwest Indiana, had paged Rivero after matching the donor with the potential recipient at UCMC. He confers with cardiologists Savitri Fedson, MD, Gene Kim, MD and UCMC’s Chief of Cardiac and Thoracic Surgery Valluvan Jeevanandam, MD—the man who will ultimately be performing the recipient’s surgery. They review the donor’s information and agree that it’s a match. They tell Rivero to “Go get it.”
The donor is generous. A hospital from Pittsburgh will take the lungs. One from Minnesota will claim the liver and kidneys. Their procurement teams are not available until later in the day. All the teams need to be there at the same time because once the procedure to remove the organs begins, there’s no stopping it. Rivero and his team will have to wait.
It’s now 4:30 p.m., and an AeroCare van’s engine idles in the UCMC Emergency Room driveway. Rivero and UCMC’s cardiac and thoracic procurement surgeon David Onsager, MD, are ready to head to Midway Airport, where they’ll board a turboprop plane that will take them to the donating hospital downstate.
But there’s a holdup because of another procurement team. They are still waiting at 5:30. The delays continue. Rivero and Onsager want to get moving.
The recipient and his family are also anxious. The call has already been made to them: It’s time. Get to the hospital. A heart just became available.
“This is the biggest problem”
Currently organ donations through transplant surgery have the potential to save an average of 79 lives every day—that’s how many people receive a transplant daily, according to the U.S. Department of Health and Human Services (HHS). Still, HHS says that about 18 people die every day waiting for an organ. Modern transplantation medicine has helped save lives since it took off more than 60 years ago with the first kidney transplant. But that success is only possible when transplantation is possible.
There are currently more than 120 million people registered as organ donors and over 123,000 people on the waiting list, according to HHS. However, these numbers are deceptive. Despite the far greater number of donors, donor doesn’t translate into useable organs.
“Demand exceeds supply,” says Michael Abecassis, MD, director of transplant services at Northwestern Memorial Hospital (NMH), founding director of the Northwestern Medicine Comprehensive Transplant Center and the former president of the American Society of Transplant Surgeons. “There are not enough organs. This is the biggest problem.”
Challenges abound. Most transplanted organs come from the deceased. Organs can only live for so long outside of the body. Kidneys can last up to 36 hours and livers up to 12—plenty of time to be sent across the country and transplanted—but the heart and lungs can only last about four hours. Many of the donors will not be good matches with those who are waiting. And there’s always the risk of organ rejection, which would put the patient back on the waiting list.
Who gets an organ when one becomes available is far more complicated than first come, first served. Wait time is considered, but there’s also organ type, age, blood type, size of the organ, the time-sensitive geographic condition and the medical urgency.
It’s not without controversy. For instance, the major transplantation network’s Liver and Intestinal Organ Transplantation Committee has said that too many livers go to people with lower risks of imminent death who are in closer proximity to the donor, rather than sending the organs farther away to save someone with a greater risk of imminent death.
Sometimes, however, even the most available organs don’t work out.
[tribulant_slideshow gallery_id=”4″]
“We have time”
Planes like this one, on missions like this one, fly on lifeguard status. The only plane that has more right-of-way in an air traffic control pattern is Air Force One. Rivero and his team even had clearance during the 9/11 groundings. The flight aboard this small twin turboprop lasts 35 minutes. It lands at 6:45 p.m. Another AeroCare van is waiting.
“Go with traffic,” Rivero tells the driver. “We have time. Code 3 on the way back.”
By 7:22 p.m., Rivero and Onsager are in scrubs waiting in the hospital’s physicians lounge. Pittsburgh and Minnesota still aren’t there.
Two hours later, all the teams have arrived, and the OR is filled with doctors and nurses, machines, sterilized tables and instruments. The donor, who suffered a brain trauma several hours ago and was pronounced brain dead, is brought in. She’s wired to more machines. They keep the dead alive. She looks no more than sedated. A checklist is reviewed to ensure that everything is in order: patient consent, blood type, brain dead. Check.
Onsager takes a look at the echocardiogram of the donor’s heart. He reaffirms that it’s good. The Pittsburgh team gets a closer look at the lungs via camera fed through the trachea. Minnesota is eager to claim the liver and kidneys.
The donor is prepped. There are decades of schooling being mumbled, directed and ordered. Before any work can begin, the room is calmed. The representative from Gift of Hope says a few words about this woman lying on the operating table: She was the mother of two young special-needs children. She was the first in her family to complete high school. She came from so little and seemed destined to have so much…
This obituary of sorts was provided by the family; likely the man with whom she shared the children and found her unconscious in their home a day before. This ceremony is a reminder that a donor is not just a body about to be stripped for parts; this donor is a person—a mother, a daughter, a friend, a lover. And because of her charity in devastating circumstances, she will go on to potentially save and improve the lives of others. This is life and death at its most real. This is humanity.
When the Gift of Hope rep is finished, the doctors and nurses are quiet with reflection. Only the gentle whirring of the machines is heard.
Then, chaos.
The teams cut and cauterize and poke and prod. Towels are stuffed inside of the body to soak up blood. There is suction. This is a complete dissection. Pittsburgh determines that the lungs are no good—there’s mild pneumonia. The liver is no good either. A phone call is made to the hospital back in Minnesota where the liver recipient is waiting. The doctor here reads off the liver’s stats once again. He hangs up the phone. It won’t do—it has more than 20 percent of fatty tissue. Minnesota will still take both kidneys and other tissues from the body to aid in research, but after the lungs and liver were turned down, there’s a general sense of disappointment in the room. Pittsburgh packs up and leaves empty-handed. The would-be recipient back East will be disappointed, too.
With the lung team out of the way, Onsager can begin working on the heart. At 11:15 p.m., he puts a cross clamp on the heart. It stops beating. The only clock that matters now has begun. From this moment on, every second is of absolute importance. This heart has until 3:15 a.m. to begin beating again in the waiting recipient’s body. Onsager infuses the heart with a solution that will help protect and preserve it on its journey back to Chicago. Then he removes it from the donor’s chest and places it in a transparent plastic container. It is placed into an ice-filled cooler. The nurses will clean up and close the donor so she can be given to her family and placed to rest. Her memory will last; her heart will face a new journey.
“We have to make better use of the kidneys we have”
With the biggest challenge being a shortage of organs, challenge number two, NMH chief of transplant Abecassis says, is that “[the recipient] has to take a lifetime of medication to prevent rejection. A third and related problem is that as good as these medications are, we still need to personalize [them] to the [individual’s] immune system.”
Pete Giannaris was born with polycystic kidney disease. By the time he was 8, cysts had hardened all around his kidneys, rendering them useless. His first transplant came from his mother. He was 9 years old. He would go on to have two more transplants—one from his father, the other from a deceased donor. His body rejected all three, putting him on dialysis and the transplant waiting list for 12 years.
“I had so many transplants that my body didn’t want a new kidney,” Giannaris says.
Organ rejection is always a risk. Our immune systems are designed to defend against foreign objects. Despite the body’s need for a new organ, that organ will be seen as an intruder. To help prevent this, doctors prescribe immunosuppressive drugs. However, these drugs come at a price. The recipient has to take a drug cocktail for the rest of his or her life, and because the drugs restrict the immune system from doing its job, there is a high probability of infection and illness.
“When people lose the transplant, the immune system often builds a rejection system of antibodies,” says Mark Stegall, MD, a kidney transplant surgeon at Mayo Clinic in Minnesota. “There was a time when people didn’t understand that.”
Giannaris contacted the Mayo Clinic after learning about its efforts to treat highly sensitized patients like him. Stegall took him on as a patient, performing Giannaris’ fourth and last kidney transplant in August 2013.
This time, the kidney transplant was successful in part because of a therapy designed to deplete the antibody-secreting cells. Before the transplant, Stegall prescribed Velcade, a drug mostly administered to multiple myeloma patients that knocks out those cells.
“I’ll have to take immunosuppressants for the rest of my life, but the amount has dropped significantly,” Giannaris says. “I hate to be one of those people who says it’s like night and day, but it’s like night and day. After 12 years of being drained, I wake up in the morning, and I’m up and running around. The best decision I ever made was to do my own research.”
Kidneys are in the highest demand. There are more than 101,000 people waiting for a kidney transplant, according to the HHS. “This number has grown dramatically since the 1980s and ’90s,” says John Friedewald, MD, associate professor of medicine and surgery at the Northwestern University Feinberg School of Medicine and a transplant nephrologist at NMH and the Kovler Organ Transplant Center. “We have to make better use of the kidneys we have.”
Better use entails options like cross-matching. This allows physicians and scientists to more accurately anticipate how a kidney transplant recipient may respond to particular cells or proteins of the donor. And there’s kidney paired donation (KPD). KPD takes medically able living donors who are incompatible with their intended recipient and pairs them up with a recipient who is compatible. For example, it’s what happens when a healthy mother wants to donate to her son, but is not a match. She is, however, a match for another woman, so the mother’s kidney will go to her. Meanwhile, the son will get a kidney from another donor who was in a similar situation.
Friedewald describes the KPD sustainability as a two-way exchange to infinity. “Better immune suppression has helped this,” he says. He admits, though, that distance does provide some limitation in allocation. “It is always easier with the other center down the street.”
[tribulant_slideshow gallery_id=”3″]
“Preparation is everything”
At 12:44 a.m., organ procurement coordinator Rivero and transplant surgeon Onsager are back in the Chicago van and heading straight to UCMC at Code 3—sirens, lights, high speed. They pull in at 12:58 a.m. The hospital hallways are a blur as they rush through with the heart in tow.
In the OR, cardiac surgeon Jeevanandam and his team are ready. There are the surgeons, nurses, an anesthesiologist and two men monitoring and managing the cardiopulmonary bypass machine, known as The Pump, which is keeping the patient alive. Two young researchers stand like eager infielders ready to receive the recipient’s old heart. This room is high-tech with large monitors on two opposing walls for all to see what’s happening. Michael Jackson plays on the Pandora station through the room’s speakers.
The patient is ready, too. He’s been under since 6:30 p.m. He is a special case. Not too long ago he had his pulmonary artery reconstructed and, therefore, had been turned down for a heart donation by another hospital because of the complexity that resulted from having open heart surgery once before. There was scar tissue in his chest, and getting him open required more work, more finesse. Despite this, UCMC took him. They often do these kinds of cases.
At 1:01 a.m., Rivero and Onsager are in scrubs again as they enter the OR. The moment they walk in with the heart, Jeevanandam begins removing the old beater. He hands it to the young researchers, and they take it to be dissected and examined.
With the heart out, Jeevanandam is surprised to see a tear in the gaping cavity. “I expected a problem with the aorta, not the area underneath,” he says. An extra half an hour of work is required to make this repair before the new heart can fill the hole. The clock keeps ticking. “If they were all the same, it’d be boring—a robot could do it.”
And remember, this is humanity.
At 2:01 a.m. Jeevanandam calls out, “First stitch.” The new heart is being sewn into place. “Preparation is everything,” he says.
Sixteen minutes later, one of the Pump techs calls out, “Plegia on!”
Plegia, short for cardioplegia, means heart paralysis. The heart can’t start pumping on its own after being empty for hours. Think of it like dry-starting a car engine without oil. When the tech calls out plegia, he’s sending oxygenated blood to the heart, which begins to warm and fill the cold and empty organ. But the blood has an arresting medication to keep the heart still, otherwise it would begin pumping on its own, and this is not ideal either. A transplanted heart has to ease into its new job. Cardioplegia is the first step in bringing the heart back to life. Bit by bit, as Jeevanandam completes each stage of connecting the heart to the body, the Pump techs add more medicated, oxygenated blood to the heart.
2:26 a.m.: “Plegia on!”
2:36 a.m.: “Plegia on!”
2:50 a.m.: “Plegia on!”
The heart is nearly full of blood but is not ready to beat on its own. Finally, at 3:04 a.m., Jeevanandam calls: “Integrate warm blood.” This will flush out the meds paralyzing the heart. But the heart doesn’t beat.
“It’s time to build made-to-measure organs”
The time may not be distant when wait lists are a thing of the past. Immunosuppressive drugs, too, may soon be as antiquated as bloodletting.
“It’s time to personalize transplantation including building made-to-measure organs,” Abecassis says.
Some 248 miles above us, at the International Space Station, such work is starting. Abba Zubair, MD, PhD, director of Cell Therapy Laboratory at Mayo Clinic in Florida, is sending human stem cells into space to see whether they grow faster. It’s microgravity he’s after.
The closest thing to microgravity we have on Earth is what occurs during pregnancy. “If you look at an embryo in a mother’s womb, the baby is suspended in fluid—it’s basically a type of microgravity,” says Zubair.
Cells need to grow three-dimensionally, which is easier to do in suspension, Zubair says. Two-dimensional growth, like what’s done here on Earth, causes the cells to flatten out due to gravity.
If the cells can be grown faster in space, and they’re safe for human use, Zubair wants to bring the cells back to Earth and use them in clinical trials. But, he says, we’re still 10 to 15 years away from growing actual organs.
“Stem cells can make tissues from all organs,” he says. “We haven’t figured out how to get the cells to grow specific to the organ, [but] if we can figure out how to differentiate, we can grow tissue.”
The best part about this is that, should the day come, the cells won’t even have to come from specific patients—anyone can use them without rejection, Zubair says. “You can collect stem cells and grow your organs in the space station to be used when needed.”
” On Bypass”
It’s 3:10 a.m., and small paddles are placed onto the still-unbeating heart.
Jeevanandam sends a skosh of electricity to kick-start it.
It faintly comes to life. This first heartbeat arrives just five minutes shy of the four-hour deadline. The entire room watches the monitors, looking for function and rhythm. Slowly, like something rising from a long, afternoon nap, the heart finds itself in its new home and begins pumping like it should; stronger, steadier… Twenty minutes later, the recipient is slowly weaned off of The Pump.
At 3:34 a.m., the Pump tech calls out, “Off bypass.” That’s the all-clear.
From here, the healing begins. The patient, a 50-year-old man with the heart of a 30-year-old woman, will have a new lease on his life and opportunities that do not come to so many others like him. And while he adjusts to his new life, much like Giannaris has, scientists and physicians and astronauts will continue to improve the way lives are saved via organ transplantation.
They will continue to blur the line between the sheer mechanics of our bodies with the overwhelming humanity of ourselves.